1. INTRODUCTION
The health of fish is adversely impacted by the contamination of aquatic ecosystems with industrial and agricultural pollutants. This influence can occur either directly through the uptake of contaminants from the water or indirectly through their diet, which may consist of smaller fish, invertebrates, and vegetation (Malik et al., 2020). The well-being and richness of fish are of significant concern due to their essential role in the diets of aquatic birds and mammals, as well as their increasing importance as a vital protein source for humans (Bernhardt & O’Connor, 2021; Zhang et al., 2024a; Li et al., 2024). Key environmental pollutants include substances that tend to accumulate in organisms, those characterized by persistence due to chemical stability or limited biodegradability (Naz et al., 2022; Pu et al., 2024), and those that exhibit high solubility, making them easily mobile in the environment (Habib et al., 2023a). Biological degradation cannot eliminate heavy metals (Tasleem et al., 2023). Exposure to higher concentrations may result in the accumulation of these metals in the organs of aquatic animals (Habib et al., 2023b). Cadmium (Cd), among the heavy metals, is recognized as a significant aquatic pollutant globally (Zheng et al., 2025). Despite being a nonessential heavy metal, it is known as one of the most potent water contaminants, capable of inducing toxicity across various levels in organisms (Sun et al., 2021). Many fish, particularly anadromous species, pass through the larval and juvenile stages in environments where they may potentially encounter sublethal concentrations of Cd (Abdel-Tawwab et al., 2024) Cd exhibits a wide array of harmful impacts on fish at sublethal concentrations. These effects manifest as bioaccumulation in crucial organs (Amouri et al., 2024), histological changes (Raeeszadeh et al., 2024), hematological effects (Fazio et al., 2022), osmoregulatory issues (Kanwal et al., 2024), impaired reproduction (Bhardwaj et al., 2024), inhibiting activities of enzymes (Jiang et al., 2024; Zhao et al., 2024), alterations in hormones (Wu et al., 2024), compromised growth, and changes in behavior (Fazio et al., 2022; Habib et al., 2024a).
Labeo rohita is a major freshwater carp species with high economic value in the aquaculture industry. Its popularity stems from its fast growth rate, adaptability to a variety of environmental conditions, and suitability for aquaculture operations (Habib et al., 2024b). As cadmium infiltrates aquatic ecosystems through anthropogenic activities (Liu et al., 2022), its sublethal effects on fish physiology merit comprehensive exploration.
Understanding how L. rohita of varying length and weight respond biochemically to Cd exposure holds implications not only for elucidating the mechanisms of metal toxicity but also for refining risk assessment models and formulating tailored conservation strategies for this ecologically significant species. The higher concentration of heavy metals renders fish susceptible to aquatic pollution, as they lack the ability to escape from the adverse effects of these contaminants (Tasleem et al., 2023). The fish biochemical parameters are crucial indicators in fish, offering significant insights into the toxicity of metals, whether acquired through their diet or from the surrounding water (Fazio et al., 2022). The main objective of this study was to assess the sublethal impact of Cd and their accumulation in various body tissues of L. rohita, considering different weight and length categories, and to analyze their effect on biochemical responses.
2. MATERIALS AND METHODS
2.1. Fish sampling and study design
The healthy L. rohita, commonly known as rohu (n=270) was purchased from the local fish hatchery in the district of Sargodha, Punjab, Pakistan. The collected samples were transported to the Research Laboratory of Animal Science (University of Lahore), where they were acclimatized for 14 days (12/12 h light/dark period) in fiberglass tanks under aerated conditions. During acclimatization, they were provided a commercial feed including crude protein (32 ± 0.6 %), crude lipid (18.32 ± 0.4 %), moisture (24.2 ± 0.9 %) and ash (13.16 ± 0.2 %). After this, the fish were transferred to the experimental tanks to undergo sublethal exposure. Fish were provided with a daily feed equivalent to 3 % of their body weight each morning and evening throughout the entire exposure period. The fish were divided in triplicates (15 fish per tank) into six groups that were control groups coded is CG1, CG2, and CG3, and their respective Cd exposure groups (CdG1, CdG2, and CdG3). The weight (g) and length (cm) measurements were as follows: CG1 (4.67 ± 0.5 g, 5.11 ± 0.22 cm) and CdG1 (4.67 ± 0.5 g, 5.11 ± 0.22 cm); CG2 (11.32 ± 0.6 g, 10.07 ± 0.54 cm) and CdG2 (11.32 ± 0.6 g, 10.07 ± 0.54 cm); and CG3 (17.42 ± 0.9 g, 15.13 ± 0.71 cm) and CdG3 (17.42 ± 0.9 g, 15.13 ± 0.71 cm). Each tank held 1000 liters of the test solution for the exposure groups and well water for the control group. The fish were sampled at different durations (1, 7, and 14 days) of Cd exposure. The tanks were oxygenated by means of air stones connected to an air compressor, ensuring thorough saturation with oxygen (Tasleem et al., 2024). The fish underwent a 24-hour period of fasting before sampling to prevent any prandial effects (Rind et al., 2023). The concentration of Cd in water samples was determined daily using inductively coupled plasma optical emission spectrometry (ICP-OES, Thermo Scientific iCAP 7000 Series). Prior to analysis, water samples were filtered through 0.45 µm membrane filters and acidified with concentrated nitric acid (HNO₃, trace metal grade) to a pH <2 to preserve metal content. Samples were then digested using a microwave digestion system with a mixture of HNO₃ and H₂O₂ following EPA Method 3015A. The instrument was calibrated using a series of certified Cd standard solutions (0.01–5.0 mg/L), and quality control was maintained by including procedural blanks and reference standards in each batch. The emission wavelength for cadmium was set at 228.802 nm, and the detection limit for Cd was approximately 0.001 mg/L. All measurements were performed in triplicate.
2.2. Sublethal Cd exposure
Prior to starting the experiment, a stock solution was prepared containing cadmium chloride of 2,000 mg·L-1 “CdCl2.2·5H2O”. Following this, the solution was appropriately diluted to reach the target nominal concentration, utilizing tank water. The selected nominal Cd concentration of 0.66 mg·L-1 had previously been recognized as a sublethal threshold for fish in our laboratory, such as 4.5 % of the determined 96-hour LC50. Throughout sublethal experiments, daily monitoring of water parameters was conducted, such as temperature (23.2 ± 0.7 °C), dissolved oxygen (5.83 ± 0.14 mg·L-1), hardness (168 ± 6.3 mg CaCO3/liter), pH (7.6 ± 0.2) and Cd (0.6302 ± 0.05 mg·L-1).
2.3. Sampling and analysis
During each designated sampling interval, 08 fish from each treatment group underwent anesthesia using a clove essence solution. Afterward, the fish were weighed, and blood samples were extracted from the caudal vein using heparinized capillaries. These blood samples were then promptly transferred to heparinized tubes, which were maintained on ice until the centrifugation process took place. Right after collecting blood, the liver, gill, intestine, and kidney tissues were meticulously dissected using sterile equipment, rinsed with physiological serum, weighed, swiftly frozen in liquid nitrogen, and subsequently stored at temperature (-80 °C) until further analysis (Habib et al., 2021). The blood samples underwent centrifugation at 10,000 rpm for 3 minutes at temperature (4 °C) to separate and collect plasma. The plasma was then aliquoted and preserved at -20 °C (Habib et al., 2023b). The homogenizer was used for liver tissue homogenization in a phosphate buffer (100 mM) of pH 7.4, 1:10, w/v. This buffer contained EDTA (2 mM) and aprotinin (150 KIU/mL), functioning as a protease inhibitor. The homogenates obtained were centrifuged using a refrigerated centrifuge at 10,000 rpm for 45 minutes at temperature (4 °C). The resulting supernatant served as the enzyme source. Briefly, the samples were subjected to cindering, followed by dissolution in highly purified HNO3 (1 mL of 65 %) (Acros Organics). The resultant solutions were then diluted to a volume of 10 mL with ultra-pure water, filtered through a 0.22 μm cellulose acetate filter, and stored in a refrigerator until subjected to metal analysis.
The levels of glucose, total protein, and cortisol were determined using test kits (Merck Sigma-Aldrich, Germany). Catalase, CAT “EC.1.11.1.6” and superoxide dismutase, SOD “EC.1.15.1.1” activities were assessed through the utilization of colorimetric assay kits in a microtiter plate format. Optical density readings were recorded using an ELISA Reader. The tests were conducted following the instructions provided in the kit inserts. The enzyme activity is defined as one unit when it facilitates the oxidation of 1 μmol substrate per minute. The outcomes are reported as units per milligram of protein. The measurement of Glutathione S-Transferase, GST “EC.2.5.1.18” activity involved tracking the generation of the reaction product between glutathione (GSH) and 1-chloro-2,4-dinitrobenzene (CDNB) at the wavelength of 340 nm (Habdous et al., 2002). GST activity was assessed by observing alterations in absorbance at 340 nm, indicative of the rate of conjugation between CDNB and GSH. The quantification of GST activity was determined by calculating the formation of micromoles of CDNB conjugate per minute per milligram of protein at a temperature (25 °C), defining one unit.
2.4. Statistical analysis
Data analysis was conducted employing statistical package programs provided by SPSS (version 22). Data of each treated and their respective control group collected at different intervals were subjected to independent samples t-test, while one-way analysis of variance, ANOVA, was applied to compare the mean among Cd treated groups. Further, the means were subsequently examined using Duncan's multiple-range tests for comparison. Statistical significance was determined by evaluating the data against a significance threshold set at P<0.05.
3. RESULTS AND DISCUSSION
Cd, being a profoundly toxic heavy metal, induces harmful effects in organisms even at low levels of exposure (Hayat et al., 2019; Akhtar et al., 2021). The study revealed no changes in water parameters or fish mortality during sublethal Cd exposure. Cd concentrations (Figure 1) in gill, intestine, and kidney tissues were significantly higher (P<0.05) in Cd-exposed groups (CdG1, CdG2, CdG3) compared to controls (CG1, CG2, CG3) across intervals (1, 7, and 14 days).
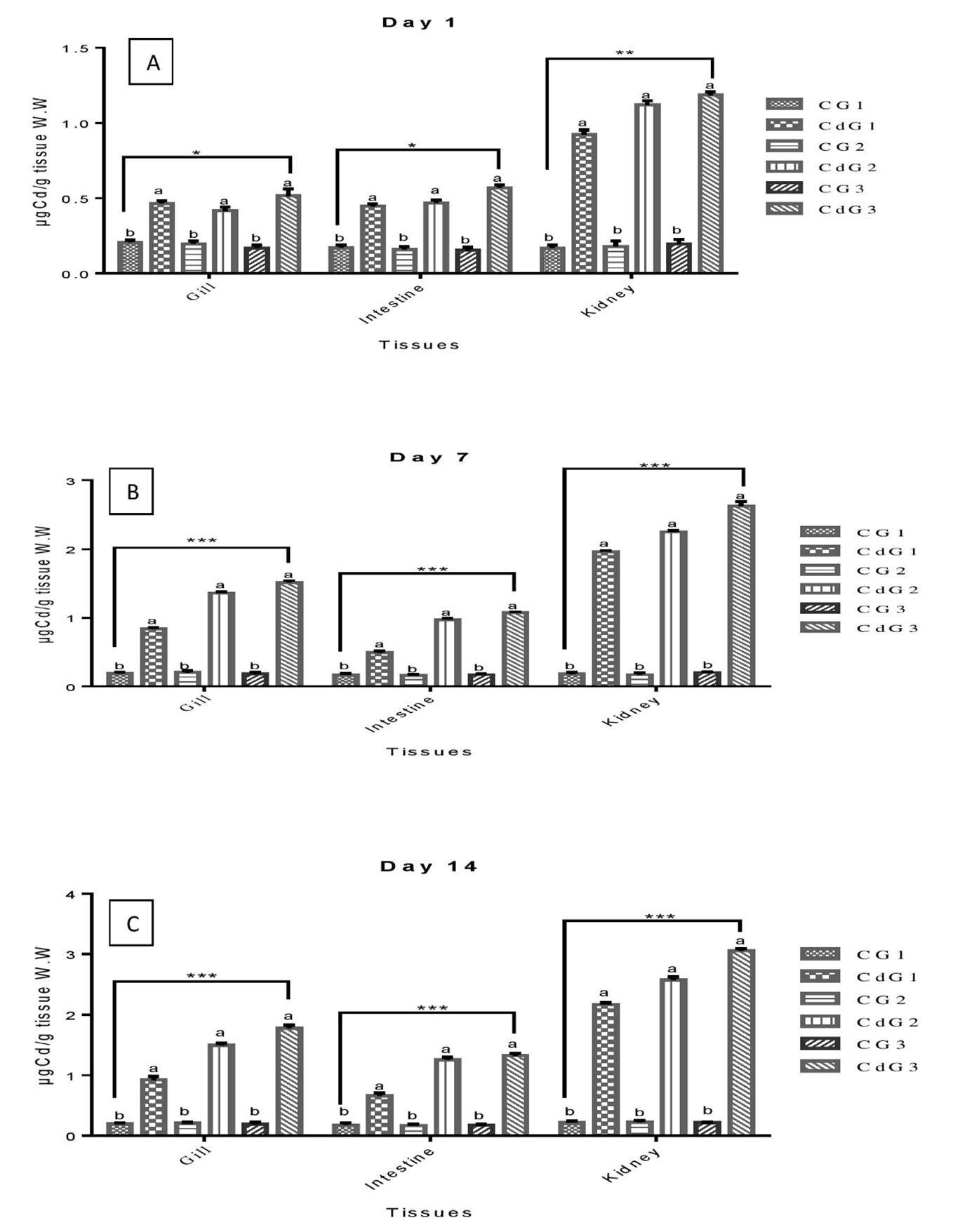
On day 1, significant Cd accumulation was observed in the gill and intestine, with the kidney showing the least significant changes. By days 7 and 14, Cd levels in all tissues were highly significant, with CdG3 consistently exhibiting the highest accumulation across intervals. The dependency of Cd accumulation on time and group aligns with previous studies showing similar patterns in waterborne metal uptake in fish tissues. For instance, Isani et al. (2009) reported higher Cd levels in the liver and kidney of Sparus aurata, Malarvizhi et al. (2017) observed Cd accumulation in Cirrhinus mrigala, and Ghedira et al. (2010) found elevated Cu and Cd levels in the liver of Sparus aurata. This study revealed tissue-specific Cd uptake in osmoregulatory tissues of L. rohita, with the kidney consistently exhibiting the highest Cd concentrations across all groups. Previous studies (for example, Hu et al. (2022) on Hexagrammos otakii, Fırat et al. (2009) on Oreochromis niloticus, and Ali et al. (2021) on Cyprinus carpio) similarly highlighted significant metal accumulation in organs like the kidney, liver, gills, and intestine under waterborne metal exposure. Cd primarily enters freshwater fish through the gills, then distributes to various organs (Moiseenko & Gashkina, 2020; Samuel et al., 2021; Naz et al., 2024a). Accumulation patterns are influenced by biotic and abiotic factors, as noted in earlier research (Alam et al., 2021; Feng et al., 2023).
CAT and SOD act in concert, forming the initial line of defense against xenobiotics that induce oxidative stress (El-Houseiny et al., 2023) Figure 2a shows CAT activity in L. rohita across Cd exposure groups (CdG1, CdG2, CdG3) at different intervals. On day 1, CAT activity was significantly higher (P<0.05) in CdG2 compared to its control. However, by days 7 and 14, CAT activity was significantly higher (P<0.05) in controls than in treated groups. Among treated groups, significant differences were observed on day 1, minimal on day 7, and highly significant (P<0.05) on day 14. CAT activity progressively decreased with exposure duration (CdG1 > CdG2 > CdG3), with the most pronounced reduction on day 14. Figure 2b illustrates SOD activity in L. rohita across Cd exposure groups (CdG1, CdG2, CdG3) at different intervals. On day 1, SOD activity was significantly higher (P<0.05) in treated groups than in controls. However, on days 7 and 14, control groups showed significantly higher (P<0.05) SOD activity compared to treated groups. Among treated groups, SOD activity was significant (P<0.05) on day 1, minimally significant on day 7, and highly significant on day 14, with a progressive decrease in activity (CdG1 > CdG2 > CdG3) over time. The focus on the liver for investigating antioxidative responses is supported by Cichoż-Lach & Michalak (2014), who identified it as the key organ reflecting antioxidant defenses. Chen et al. (2023) observed notable CAT and SOD activities in the liver of Oreochromis niloticus, while Taysi (2024) reported significant changes in liver CAT activity after Cd exposure. This study aligns with prior findings, showing significant alterations in hepatic SOD levels during Cd exposure (Hu et al., 2021; Zhang et al., 2024b). The observed decline in SOD activity may result from oxidative stress induced by Cd or its direct interaction with the enzyme, as noted by Cheng et al (2022). Cd is known to enhance reactive oxygen species (ROS) production and inhibit certain antioxidative enzymes (Sachdev et al., 2021; Zandi & Schnug, 2022).
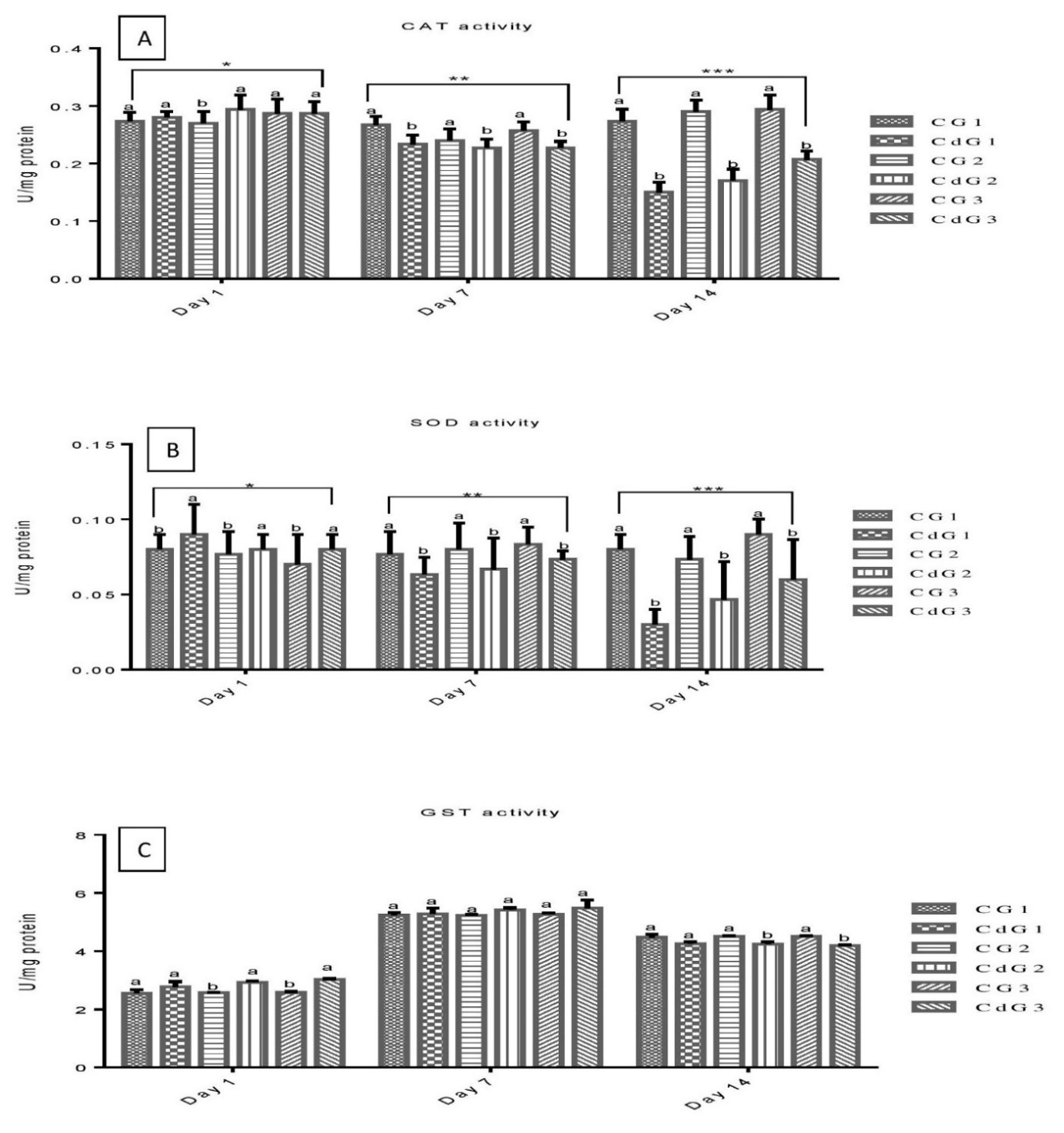
GSTs play a vital role in the cellular antioxidant defense system, facilitating the conjugation of GSH to diverse electrophilic compounds. This process contributes to cellular detoxification (Demirci-Cekic et al., 2022) Figure 2C shows GST activity in L. rohita across Cd exposure groups. On day 1, CdG2 and CdG3 had significantly higher (P<0.05) activity than controls. By day 7, no significant differences were observed between treated and control groups. On day 14, control groups showed significantly higher (P<0.05) GST activity than treated groups. No significant differences were found among treated groups across all intervals. Le Thu et al. (2021) reported no significant changes in hepatic GST activity in fish exposed to waterborne Cd for 28 days. In contrast, de Farias Araujo et al. (2022) observed increased GST activity in O. niloticus following Cd exposure.
Table 1 summarizes the biochemical parameters of L. rohita across three groups based on length and weight under Cd exposure for 1, 7, and 14 days. No significant differences (P<0.05) in liver protein levels were observed between treated and control groups or among treated groups on days 1 and 7. However, by day 14, liver protein levels were significantly higher (P<0.05) in treated groups compared to controls, with CdG1 and CdG2 showing the highest values. Fish utilize glucose as an energy substrate during stressful conditions, and prior studies have indicated that Cd induces hyperglycemia (Gashkina, 2024; Chakraborty et al., 2024). However, this response typically terminates within a few days (Fazio et al., 2022). Ya et al. (2021) emphasized that Cd could cause liver damage and disrupt glucose homeostasis. In this study the glucose levels in each Cd exposure group were significantly higher (P<0.05) than those in their control at different intervals. Moreover, within the treated groups, CdG1 exhibited notably higher (P<0.05) glucose levels compared to the others on days 1, 7, and 14. Conversely, CdG3 showed a consistently lower mean concentration of glucose. The Cd has the capability to cause osmo-ionic disturbances and trigger the hypothalamic–pituitary, leading to the secretion of cortisol (Naz et al., 2024b), resulting in the metabolism of carbohydrates and the onset of hyperglycemia (Zhao et al., 2022). Plasma protein levels significantly increased (P<0.05) in all Cd exposure groups compared to controls at all intervals, with CdG1 showing the highest level on day 14.
|
Table 1. Biochemical parameters of L. rohita exposed to Cd at different intervals. |
||||||
|
Parameters |
CG1 |
CdG1 |
CG2 |
CdG2 |
CG3 |
CdG3 |
|
Day 1 |
||||||
|
Liver protein, mg/mg |
0.127 ± 0.002a |
0.131 ± 0.001aA |
0.125 ± 0.004a |
0.129± 0.003aA |
0.126 ± 0.002a |
0.128 ± 0.006aA |
|
Glucose, mg/dl |
51.34 ± 3.02b |
68.32 ± 4.13aA |
53.26 ± 2.07b |
65.12 ± 3.18aAB |
49.09 ± 2.57b |
63.54 ± 4.06aB |
|
Plasma protein, g/dl |
2.08 ± 0.05b |
2.67 ± 0.18aA |
2.06 ± 0.03b |
2.74 ± 0.23aA |
2.12 ± 0.07b |
2.64 ± 0.13aA |
|
Cortisol, ng/l |
15.04 ± 1.25b |
38.12 ± 2.32aA |
17.24 ± 1.58b |
32.53 ± 2.35aAB |
14.89 ± 1.02b |
31.48 ± 1.48aB |
|
Day 7 |
||||||
|
Liver protein, mg/mg |
0.132 ± 0.004a |
0.142 ± 0.007aA |
0.129 ± 0.002a |
0.138 ± 0.006aA |
0.132 ± 0.003a |
0.144 ± 0.003aA |
|
Glucose, mg/dl |
44.12 ± 4.56b |
69.12 ± 2.13aA |
42.54 ± 2.68b |
66.32 ± 2.64aAB |
46.75 ± 4.82b |
63.32 ± 3.32aB |
|
Plasma protein, g/dl |
2.04 ± 0.42b |
2.45 ± 0.34aA |
2.07 ± 0.83b |
2.49 ± 0.53aA |
2.11 ± 0.08b |
2.48 ± 0.28aA |
|
Cortisol, ng/l |
16.17 ± 1.23b |
28.72 ± 1.04aA |
14.19 ± 1.29b |
25.93 ± 1.32aAB |
15.26 ± 1.13b |
23.28 ± 1.12aB |
|
Day 14 |
||||||
|
Liver protein, mg/mg |
0.138 ± 0.004b |
0.154 ± 0.05aA |
0.132 ± 0.002b |
0.149 ± 0.07aA |
0.131 ± 0.001b |
0.142 ± 0.05aB |
|
Glucose, mg/dl |
54.32 ± 2.48b |
67.32 ± 4.75aA |
52.56 ± 2.15b |
64.04 ± 3.12aB |
55.28 ± 2.29b |
60.13 ± 3.11aC |
|
Plasma protein, g/dl |
2.32 ± 0.27b |
2.78 ± 0.56aA |
2.29 ± 0.16b |
2.73 ± 0.39aAB |
2.34 ± 0.19b |
2.69 ± 0.23aB |
|
Cortisol, ng/l |
18.12 ± 0.87b |
33.32 ± 1.45aA |
16.43 ± 0.62b |
31.36 ± 1.63aAB |
19.74 ± 0.21b |
29.42 ± 1.29aB |
|
Note: in a single row, the use of different superscripts in small letters indicates a significant difference between the control and treated groups, while the different superscripts in capital letters represent a significant difference among the Cd treated groups. |
||||||
Cortisol levels were significantly elevated (P<0.05) in all treated groups compared to controls across 1, 7, and 14 days, with CdG1 consistently showing the highest levels among treated groups. According to the study of Öner et al. (2008), the protein level in O. nilotcius increases after exposure to Ag for 30 days duration. Almeida et al. (2001) reported a decreased level of liver and muscle protein of O. niloticus after being treated with Cd. It is important to emphasize that a consistent pattern in tissue protein alterations during waterborne Cd exposure has not been universally established across various studies (Fazio et al., 2022).
4. CONCLUSIONS
The 14-day sublethal exposure of L. rohita to 0.66 mg·L-1 of Cd resulted in significant physiological stress, as evidenced by elevated levels of oxidative stress markers, altered antioxidant enzyme activities (such as SOD, CAT, and GPx), and disrupted metabolic function. A clear size-dependent pattern of Cd bioaccumulation was observed, with smaller fish accumulating higher concentrations of Cd, particularly in the liver and gills, suggesting greater vulnerability due to higher metabolic rates and surface area-to-volume ratios. The observed biochemical responses indicate an attempt at physiological adaptation, yet these mechanisms were insufficient to fully counteract the oxidative damage. These findings highlight not only the toxicological risk posed by Cd in freshwater environments but also the importance of considering organism size and organ specificity in ecological risk assessments. Consequently, the study underscores the urgent need for stringent monitoring and regulation of heavy metal pollutants to safeguard aquatic life and associated human health risks through the food chain.
Acknowledgements
The authors acknowledge and appreciate the Ongoing Research Funding Program (ORF-2025-758), King Saud University, Riyadh, Saudi Arabia.
Funding
This research was supported by the Ongoing Research Funding Program (ORF-2025-758) at King Saud University in Riyadh, Saudi Arabia.
REFERENCES
- Abdel-Tawwab, M., Eldessouki, E.A., Abd-Ellatief, H.A., Khalil, R.H., El-Sabbagh, N.M., Saleh, H.M., Saleh, N.A., Abdelhakim, T. & Samak, D.H., 2024. Antagonistic effects of Bacillus subtilis-derived chitosan nanoparticles on growth performance, stress biomarkers, and histological alterations of cadmium-intoxicated Nile tilapia fingerlings. Aquaculture International, 32(7), 10269–10299.
- Akhtar, N., Syakir Ishak, M.I., Bhawani, S.A. & Umar, K., 2021. Various natural and anthropogenic factors responsible for water quality degradation: A review. Water, 13(19), 2660.
- Alam, R.T., Abu Zeid, E.H., Khalifa, B.A., Arisha, A.H. & Reda, R.M., 2021. Dietary exposure to methyl mercury chloride induces alterations in hematology, biochemical parameters, and mRNA expression of antioxidant enzymes and metallothionein in Nile tilapia. Environmental Science and Pollution Research, 28, 31391–31402.
- Ali, Z., Yousafzai, A.M., Sher, N., Muhammad, I., Nayab, G.E., Aqeel, S.A.M., Shah, S.T., Aschner, M., Khan, I. & Khan, H., 2021. Toxicity and bioaccumulation of manganese and chromium in different organs of common carp (Cyprinus carpio). Toxicology Reports, 8, 343–348.
- Almeida, J.A., Novelli, E.L.B., Silva, M.D.P. & Júnior, R.A., 2001. Environmental cadmium exposure and metabolic responses of the Nile tilapia, Oreochromis niloticus. Environmental Pollution, 114(2), 169–175.
- Amouri, R.E., Ujan, J.A., Raza, A., Mushtaq, A., Saeed, M.Q., Masud, S., Habib, S.S., Milošević, M., Al-Rejaie, S.S. & Mohany, M., 2024. Sublethal concentrations of cadmium and lead: effects on hemato-biochemical parameters and tissue accumulation in Wallagu attu. Biological Trace Element Research, 203(1), 400–410.
- Bernhardt, J.R. & O’Connor, M.I., 2021. Aquatic biodiversity enhances multiple nutritional benefits to humans. Proceedings of the National Academy of Sciences, 118(15), e1917487118.
- Bhardwaj, J.K., Siwach, A., Sachdeva, D. & Sachdeva, S.N., 2024. Revisiting cadmium-induced toxicity in the male reproductive system: an update. Archives of Toxicology, 98, 3619–3639.
- Chakraborty, P., Krishnani, K.K., Mulchandani, A., Paniprasad, K., Sarkar, D.J., Sawant, P.B., Kumar, N., Sarkar, B., Mallik, A., Pal, P. & Nagendrasai, K., 2024. Speciation-specific chromium bioaccumulation and detoxification in fish using hydrogel microencapsulated biogenic nanosilver and zeolite synergizing with biomarkers. Environmental Geochemistry and Health, 46(8), 298.
- Chen, S., Zhang, Y., Wei, Y., Guo, Q. & Gan, L., 2023. Acetylcholinesterase activity, histopathological changes, lipid peroxidation, and stress-related gene expression in Nile tilapia (Oreochromis niloticus) exposed to waterborne methidathion. Aquaculture Reports, 33, 101757.
- Cheng, C., Ma, H., Liu, G., Fan, S. & Guo, Z., 2022. Mechanism of cadmium exposure-induced hepatotoxicity in the mud crab (Scylla paramamosain): Activation of oxidative stress and Nrf2 signaling pathway. Antioxidants, 11(5), 978.
- Cichoż-Lach, H. & Michalak, A., 2014. Oxidative stress as a crucial factor in liver diseases. World Journal of Gastroenterology: WJG, 20(25), 8082.
- de Farias Araujo, G., Soares, L.O.S., Junior, S.F.S., De Carvalho, L.V.B., Rocha, R.C.C., Saint'Pierre, T., Hauser-Davis, R.A., Correia, F.V. & Saggioro, E.M., 2022. Oxidative stress and metal homeostasis alterations in Danio rerio (zebrafish) under single and combined carbamazepine, acetamiprid, and cadmium exposures. Aquatic Toxicology, 245, 106122.
- Demirci-Cekic, S., Özkan, G., Avan, A.N., Uzunboy, S., Çapanoğlu, E. & Apak, R., 2022. Biomarkers of oxidative stress and antioxidant defense. Journal of Pharmaceutical and Biomedical Analysis, 209, 114477.
- El-Houseiny, W., Anter, R.G., Arisha, A.H., Mansour, A.T., Safhi, F.A., Alwutayd, K.M., Elshopakey, G.E., Abd El-Hakim, Y.M. & Mohamed, E.M., 2023. Growth retardation, oxidative stress, immunosuppression, and inflammatory disturbances induced by herbicide exposure of catfish (Clarias gariepinus) and the alleviation effect of dietary wormwood (Artemisia cina). Fishes, 8(6), 297.
- Fazio, F., Habib, S.S., Naz, S., Hashmi, M.A.H., Saoca, C. & Ullah, M., 2022. Cadmium sub-lethal concentration effect on growth, haematological and biochemical parameters of Mystus seenghala (Sykes, 1839). Biological Trace Element Research, 200(5), 2432–2432.
- Feng, S., Cao, M., Tang, P., Deng, S., Chen, L., Tang, Y., Zhu, L., Chen, X., Huang, Z., Shen, M. & Yang, F., 2023. Microcystins Exposure Associated with Blood Lipid Profiles and Dyslipidemia: A Cross-Sectional Study in Hunan Province, China. Toxins, 15(4), 293.
- Fırat, Ö., Cogun, H.Y., Aslanyavrusu, S. & Kargın, F., 2009. Antioxidant responses and metal accumulation in tissues of Nile tilapia Oreochromis niloticus under Zn, Cd, and Zn+Cd exposures. Journal of Applied Toxicology, 29(4), 295–301.
- Gashkina, N.A., 2024. Metal toxicity: Effects on energy metabolism in fish. International Journal of Molecular Sciences, 25(9), 5015.
- Ghedira, J., Jebali, J., Bouraoui, Z., Banni, M., Guerbej, H. & Boussetta, H., 2010. Metallothionein and metal levels in liver, gills, and kidney of Sparus aurata exposed to sublethal doses of cadmium and copper. Fish Physiology and Biochemistry, 36(1), 101–107.
- Habdous, M., Vincent-Viry, M., Visvikis, S. & Siest, G., 2002. Rapid spectrophotometric method for serum glutathione S-transferases activity. Clinica Chimica Acta, 326(1-2), 131–142.
- Habib, S.S., Batool, A.I., Rehman, M.F.U. & Naz, S., 2023a. Comparative analysis of hemato‐biochemical profile, growth performance, and body composition of common carp cultured under different feed and habitats (biofloc technology and earthen pond systems). North American Journal of Aquaculture, 85(3), 277–290.
- Habib, S.S., Batool, A.I., Rehman, M.F.U. & Naz, S., 2023b. Evaluation and association of heavy metals in commonly used fish feed with metals concentration in some tissues of O. niloticus cultured in biofloc technology and earthen pond system. Biological Trace Element Research, 201(6), 3006–3016.
- Habib, S.S., Fazio, F., Naz, S., Arfuso, F., Piccione, G., Rehman, H.U., Achakzai, W.M., Uddin, M.N., Rind, K.H. & Rind, N.A., 2021. Seasonal variations in haematological parameters and body composition of Labeo rohita (Rohu) and Cirrhinus mrigala (Mrigal carp) in River Indus, District Dera Ismail Khan, Pakistan. Turkish Journal of Fisheries and Aquatic Sciences, 21(9), 435–441.
- Habib, S.S., Maqaddas, S., Fazio, F., Amouri, R.E., Shaikh, G.S., Rahim, A., Khan, K., Ullah, J., Mohany, M., Parrino, V. & Al-Eman, A., 2024a. Evaluation of lead exposure effects on tissue accumulation, behavior, morphological and hemato-biochemical changes in common carp (Cyprinus carpio). Journal of Trace Elements in Medicine and Biology, 86, 127523.
- Habib, S.S., Naz, S., Saeed, M.Q., Ujan, J.A., Masud, S., Mushtaq, A., Ullah, M., Khan, K., Zahid, M., Al-Rejaie, S.S. & Mohany, M., 2024b. Assessment of heavy metal levels in polyculture fish farms and their aquatic ecosystems: an integrative study addressing environmental and human health risks associated with dam water usage. Environmental Geochemistry and Health, 46(8), 267.
- Hayat, M.T., Nauman, M., Nazir, N., Ali, S. & Bangash, N., 2019. Environmental hazards of cadmium: Past, present, and future. In Cadmium toxicity and tolerance in plants, 163–183. Academic Press.
- Hu, F., Sun, M., Li, L., Gao, F., Jian, Y., Wang, X. & Guo, W., 2022. Effects of environmental cadmium on cadmium accumulation, oxidative response, and microelements regulation in the liver and kidney of Hexagrammos otakii. Journal of Ocean University of China, 21(2), 479–485.
- Hu, F., Yin, L., Dong, F., Zheng, M., Zhao, Y., Fu, S., Zhang, W. & Chen, X., 2021. Effects of long-term cadmium exposure on growth, antioxidant defense, and DNA methylation in juvenile Nile tilapia (Oreochromis niloticus). Aquatic Toxicology, 241, 106014.
- Isani, G., Andreani, G., Cocchioni, F., Fedeli, D., Carpené, E. & Falcioni, G., 2009. Cadmium accumulation and biochemical responses in Sparus aurata following sub-lethal Cd exposure. Ecotoxicology and Environmental Safety, 72(1), 224–230.
- Jiang, H., Li, R., Zhao, M., Peng, X., Sun, M., Liu, C., Liu, G. & Xue, H., 2024. Toxic effects of combined exposure to cadmium and diclofenac on freshwater crayfish (Procambarus clarkii): Insights from antioxidant enzyme activity, histopathology, and gut microbiome. Aquatic Toxicology, 268, 106844.
- Kanwal, H., Shoaib, I., Noman, A., Maqsood, M.F., Naheed, R., Alzoub, O., Hashem, M., Elnour, R., Alzuaibr, F.M., Khalid, N. & Irshad, M.K., 2024. Biostimulant-mediated cellular repair by improving antioxidant dynamics and osmoregulation against metal stress in canola. Turkish Journal of Agriculture and Forestry, 48(4), 580–594.
- Le Thu, H., Bui, T.H. & Pham, T.D., 2021. Effect of heavy metals on the activity of catalase and glutathione-S-transferase in Nile tilapia fish (Oreochromis niloticus). VNU Journal of Science: Natural Sciences and Technology, 37(4), 82-87.
- Li, Y., He, X., Sun, B., Hu, N., Li, J., You, R., Tao, F., Fang, L., Li, Y. & Zhai, Q., 2024. Combined exposure of beta-cypermethrin and emamectin benzoate interferes with the HPO axis through oxidative stress, causing an imbalance of hormone homeostasis in female rats. Reproductive Toxicology, 123, 108502.
- Liu, Y., Chen, Q., Li, Y., Bi, L., Jin, L. & Peng, R., 2022. Toxic effects of cadmium on fish. Toxics, 10(10), 622.
- Malarvizhi, A., Saravanan, M., Poopal, R.K., Hur, J.H. & Ramesh, M., 2017. Accumulation of cadmium and antioxidant and hormonal responses in the Indian major carp Cirrhinus mrigala during acute and sublethal exposure. Water, Air, & Soil Pollution, 228, 310.
- Malik, D.S., Sharma, A.K., Sharma, A.K., Thakur, R. & Sharma, M., 2020. A review on the impact of water pollution on freshwater fish species and their aquatic environment. Advances in Environmental Pollution Management: Wastewater Impacts and Treatment Technologies, 1, 10–28.
- Moiseenko, T.I. & Gashkina, N.A., 2020. Distribution and bioaccumulation of heavy metals (Hg, Cd, and Pb) in fish: Influence of the aquatic environment and climate. Environmental Research Letters, 15(11), 115013.
- Naz, S., Arshad, M., Majeed, S., Maqaddas, S., Habib, S.S., Kesbiç, O.S., Al-Rejaie, S.S., Mohany, M., Bottari, T., Aragona, F. & Fazio, F., 2024a. Assessing heavy metal contamination in commonly used fertilizers for polyculture fish ponds and its implications for human health: A comprehensive investigation. Biological Trace Element Research, 203(6), 3318-3334.
- Naz, S., Fazio, F., Habib, S.S., Nawaz, G., Attaullah, S., Ullah, M., Hayat, A. & Ahmed, I., 2022. Incidence of heavy metals in the application of fertilizers to crops (wheat and rice), a fish (common carp) pond and a human health risk assessment. Sustainability, 14(20), 13441.
- Naz, S., Rind, K.H., Afzal, W., Ujan, J.A. & Mohany, M., 2024b. Heavy metal pollution in commonly consumed fish species: Seasonal variations and health risks. Journal of Environmental Science and Health, Part B, 59(10), 678–685.
- Öner, M., Atli, G. & Canli, M., 2008. Changes in serum biochemical parameters of freshwater fish Oreochromis niloticus following prolonged metal (Ag, Cd, Cr, Cu, Zn) exposures. Environmental Toxicology and Chemistry: An International Journal, 27(2), 360–366.
- Pu, X., Sheng, S., Fu, Y., Yang, Y. & Xu, G., 2024. Construction of circRNA–miRNA–mRNA ceRNA regulatory network and screening of diagnostic targets for tuberculosis. Annals of Medicine, 56(1), 2416604.
- Raeeszadeh, M., Moradian, M., Khademi, N. & Amiri, A.A., 2024. The effectiveness of time in treatment with vitamin C and broccoli extract on cadmium poisoning in mice: histological changes of testicular tissue and cell apoptotic index. Biological Trace Element Research, 202(7), 3278–3292.
- Rind, K.H., Habib, S.S., Ujan, J.A., Fazio, F., Naz, S., Batool, A.I., Ullah, M., Attaullah, S., Khayyam, K. & Khan, K., 2023. The effects of different carbon sources on water quality, growth performance, hematology, immune, and antioxidant status in cultured Nile Tilapia with biofloc technology. Fishes, 8(10), 512.
- Sachdev, S., Ansari, S.A., Ansari, M.I., Fujita, M. & Hasanuzzaman, M., 2021. Abiotic stress and reactive oxygen species: Generation, signaling, and defense mechanisms. Antioxidants, 10(2), 277.
- Samuel, M.S., Datta, S., Khandge, R.S. & Selvarajan, E., 2021. A state-of-the-art review on characterization of heavy metal-binding metallothionein proteins and their widespread applications. Science of the Total Environment, 775, 145829.
- Sun, Y., Zong, C., Liu, J., Zeng, L., Li, Q., Liu, Z., Li, Y., Zhu, J., Li, L., Zhang, C. & Zhang, W., 2021. C-myc promotes miR-92a-2-5p transcription in rat ovarian granulosa cells after cadmium exposure. Toxicology and Applied Pharmacology, 421, 115536.
- Tasleem, S., Alotaibi, B.S., Masud, S., Habib, S.S., Acar, Ü., Gualandi, S.C., Ullah, M., Khan, K., Fazio, F. & Khayyam, K., 2024. Biofloc system with different carbon sources improved growth, haematology, nonspecific immunity, and resistivity against Aeromonas hydrophila in common carp (Cyprinus carpio). Aquaculture Research, 2024(1), 7652354.
- Tasleem, S., Masud, S., Habib, S.S., Naz, S., Fazio, F., Aslam, M., Ullah, M. & Attaullah, S., 2023. Investigation of the incidence of heavy metals contamination in commonly used fertilizers applied to vegetables, fish ponds, and human health risk assessments. Environmental Science and Pollution Research, 30(45), 100646–100659.
- Taysi, M.R., 2024. Assessing the effects of cadmium on antioxidant enzymes and histological structures in rainbow trout liver and kidney. Scientific Reports, 14(1), 27453.
- Wu, Y., Chen, L., Yan, X., Xiao, J., Ma, Z., Tang, Z., Guo, Z., Li, L., Tong, G., Tan, H. & Chen, F., 2024. The effect of copper–cadmium co-exposure and hormone remediation on the ovarian transcriptome of Nile Tilapia (Oreochromis niloticus). Fishes, 9(2), 67.
- Ya, J., Xu, Y., Wang, G. & Zhao, H., 2021. Cadmium induced skeletal underdevelopment, liver cell apoptosis, and hepatic energy metabolism disorder in Bufo gargarizans larvae by disrupting thyroid hormone signaling. Ecotoxicology and Environmental Safety, 211, 111957.
- Zandi, P. & Schnug, E., 2022. Reactive oxygen species, antioxidant responses, and implications from a microbial modulation perspective. Biology, 11(2), 155.
- Zhang, Y., Zhang, X., Cao, D., Yang, J., Mao, H., Sun, L. & Wang, C., 2024a. Integrated multi-omics reveals the relationship between growth performance, rumen microbes and metabolic status of Hu sheep with different residual feed intakes. Animal Nutrition, 18, 284-295.
- Zhang, Q., Xie, Y., Qin, R., Huang, E., Zhang, Z., Zhou, J., Liu, D., Meng, L., Liu, Y. & Tong, T., 2024b. Effects of cadmium on the growth, muscle composition, digestion, gene expression of antioxidant and lipid metabolism in juvenile tilapia (Oreochromis niloticus). Frontiers in Marine Science, 11, 1443484.
- Zhao, L., Liao, L., Tang, X., Liang, J., Liu, Q., Luo, W., Adam, A.A., Luo, J., Li, Z., Yang, S. & Rahimnejad, S., 2022. High-carbohydrate diet altered conversion of metabolites and deteriorated health in juvenile largemouth bass. Aquaculture, 549, 737816.
- Zhao, L., Liao, M., Li, L., Chen, L., Zhang, T. & Li, R., 2024. Cadmium activates the innate immune system through the AIM2 inflammasome. Chemico-Biological Interactions, 399, 111122.
- Zheng, R., Zhu, J., Liao, P., Wang, D., Wu, P., Mao, W., Zhang, Y. & Wang, W., 2025. Environmental colloid behaviors of humic acid-Cadmium nanoparticles in aquatic environments. Journal of Environmental Sciences, 149, 663–675.
